| AIDScience Vol. 2, No. 8, 26 April 2002 |
| Development of the dual-subtype feline immunodeficiency virus vaccine |
| By Janet K. Yamamoto, Barbara A. Torres and Ruiyu Pu |
| Department of Pathobiology, College of Veterinary Medicine, University of Florida, Gainesville, Florida 32611, United States |
| Address correspondence to: YamamotoJ@mail.vetmed.ufl.edu |
Abstract
![]() eline immunodeficiency virus (FIV) is a natural infection of domestic cats that results in an immunodeficiency syndrome resembling HIV infection in humans. Since its discovery in 1987, FIV infection of cats has been used in vaccine studies as a small-animal model of human AIDS. Recent FIV vaccine data are compared as to vaccine approach, vaccine strains used, immunity, efficacy, and challenge strains. Also discussed are the design and approach of the first animal lentivirus vaccine to be approved, the dual-subtype FIV vaccine. This highly efficacious vaccine, developed by researchers at the University of Florida, uses viruses from two subtypes of FIV that were isolated from long-term nonprogressor cats. The criteria for vaccine virus selection is discussed, as well as the approach of using inactivated whole virus, which is not the currently preferred vaccine design for HIV.
eline immunodeficiency virus (FIV) is a natural infection of domestic cats that results in an immunodeficiency syndrome resembling HIV infection in humans. Since its discovery in 1987, FIV infection of cats has been used in vaccine studies as a small-animal model of human AIDS. Recent FIV vaccine data are compared as to vaccine approach, vaccine strains used, immunity, efficacy, and challenge strains. Also discussed are the design and approach of the first animal lentivirus vaccine to be approved, the dual-subtype FIV vaccine. This highly efficacious vaccine, developed by researchers at the University of Florida, uses viruses from two subtypes of FIV that were isolated from long-term nonprogressor cats. The criteria for vaccine virus selection is discussed, as well as the approach of using inactivated whole virus, which is not the currently preferred vaccine design for HIV.
Introduction
Discovered in 1987, feline immunodeficiency virus (FIV) infects domestic cats and causes an immunodeficiency disease resembling HIV-1 infection of humans. Feline AIDS is an important immunological disease of domestic cats and is marked by a significant loss of CD4+ T cells followed by the manifestation of an immunodeficiency syndrome. Although FIV is the most recently recognized member of the lentiviral subfamily (1), it has existed longer in domestic cats than either of its relatives, HIV-1 or HIV-2, both of which are thought to have emerged as a recent xenoinfection of humans with SIVcpz and SIVsm, respectively, from African monkeys (2). Consequently, FIV has less genetic intra- and intersubtype diversity than HIV-1 subtypes have. At least five FIV clades have been identified, whereas HIV-1 is classified into three major groups, M (Major), N (for New, or non-M, non-O), and O (Outlier) (3-5). The majority of HIV-1 isolates belong to the M group, which consists of at least nine subtypes or clades. Mutations leading to quasi-species formation within a cat are more limited than HIV-1 variant development within a person (6). Furthermore, the natural transmission dose for FIV may be lower than for HIV-1, because the natural transmission route (biting) limits the rate of FIV transmission (7, 8).
Vaccines that use inactivated viruses as antigens are considered to be conventional vaccines, and this approach is the second most commonly used in veterinary and human medicine (9, 10). In veterinary medicine, inactivated viruses have been used for feline leukemia virus (FeLV), a mammalian type C retrovirus belonging to the Retroviridae family. A concern in using this approach in humans is the possibility of active infection due to incomplete inactivation of the vaccine virus. However, there have been no reports of infection due to inadequately inactivated FeLV vaccine virus.
Developing a vaccine that confers protection against worldwide isolates is clearly a major challenge whether it is a vaccine for FIV or HIV-1. However, a vaccine against FIV, rather than HIV-1, is more plausible using the technology currently available for the following reasons: 1) inter- and intrasubtype variation is lower for FIV than for HIV-1, 2) fewer subtypes have been identified for FIV than for HIV-1, whose numbers are still growing due to its relatively recent emergence in humans via xenoinfection, 3) the rate of mutation within an infected host is lower for FIV than HIV-1, 4) the transmission dose as well as the frequency of exposure may be lower for FIV than for HIV-1, and 5) the use of inactivated virus vaccines is more readily acceptable in veterinary medicine where precedent has been set by the FeLV vaccines.
FIV vaccine trials
FIV vaccine successes and failures have been observed with both conventional and molecular FIV vaccines. Throughout the 14 years of FIV vaccine trials, three fundamental questions were constantly raised: 1) what criteria define the optimal vaccine virus strains, 2) what is the best approach to develop a vaccine that confers protection against worldwide isolates, and 3) how to design vaccine trials that translate protection against experimental challenge to prophylaxis against natural infection.
FIV strains characterized by different laboratories were used as vaccine viruses in initial FIV vaccine trials. Some of these vaccines protected against homologous challenges (same vaccine strain) but were ineffective against distinctly heterologous strains, such as those from different subtypes. Subunit vaccine designs failed most frequently, whereas vaccines containing whole virion structural proteins or genes provided some protection (11, 12). Many of these vaccines were compared against a vaccine based on the first FIV isolate, FIVPET. The FIVPET vaccine was able to elicit homologous protection in multiple laboratories. Careful analysis of the results from different FIV vaccine trials indicated the inability of single-subtype FIV vaccine to protect cats against distinctly heterologous FIV strains including those from other subtypes. As a result, various formulations of dual- and triple-subtype FIV vaccines were designed and refined until arriving at a dual-subtype FIV vaccine formulation that is the prototype of the FIV vaccine recently approved by the U.S. Department of Agriculture. The USDA-approved FIV vaccine is a conventional inactivated vaccine consisting of FIV isolates from the United States (subtype A) and Asia (subtype D) (13).
| Table 1. [Enlarge] Selection of FIV vaccine strains. |
 |
Selection of vaccine strain(s) and vaccine design. The selection of vaccine virus(es) was initially arbitrary and involved using strains that were isolated by different laboratories. This approach is still in practice in some of the laboratories currently developing FIV vaccines, whereas a number of laboratories have extended collaborations and performed experimental vaccine trials using multiple vaccine strains and designs (11, 12, 14). Eight vaccine strains are most commonly used in experimental FIV vaccine trials (Table 1). Five of the eight vaccine strains have the env gene sequence of subtype A, two are from subtype B, and the last is from subtype D. Based on our current understanding of the global distribution of FIV, the use of subtype A and B strains as vaccine antigens is in agreement with the global distribution of FIV, where the predominant worldwide subtypes are A and B, followed by C, D, and E (Figure 1).
Recent findings suggest that HIV-infected individuals generally develop antibodies that neutralize viruses from the same subtype as the one infecting them, but some have been shown to develop antibodies that neutralize strains from other subtypes. A similar observation has been reported for FIV (4). Although genetic subtyping does not always correlate with neutralizing serotyping, the former provides a logical scheme for selecting vaccine strain(s) that may induce broader immunity, such as virus-neutralizing antibodies (VNA). Experimental vaccine trials using these vaccine strains have identified several vaccine designs that conferred protection against homologous challenge (Table 2).
| Table 2. [Enlarge] Years 1988-2002a summary of experimental FIV vaccines. |
 |
However, protection against distinctly heterologous in vivo-derived challenges has been more difficult to achieve using single-subtype vaccines. In vivo-derived inocula consist of either pooled plasma or leukocytes taken from cats infected with FIV strains that were passaged in vivo. To date, only conventional vaccine designs have successfully protected against distinctly heterologous in vivo-derived strains (15, 16).
Recently, interesting results were obtained in a natural-contact study comparing the infection rates between unvaccinated cats and cats vaccinated with a vaccine consisting of inactivated subtype B FIV-infected cells. All vaccinated cats were protected (infection rate of 0/12), whereas the unvaccinated group had an infection rate of 5/14 cats when both groups were placed in a free-roaming shelter for 22 months with naturally FIV-infected cats (16). The vaccinated group received six immunizations at 0, 3, 16, 40, and 60 weeks. All five infected control cats were infected with subtype B FIV isolates, suggesting that the vaccine protected against contact challenges with homologous subtype. These investigators have reported that a major limitation of this vaccine design is short duration of vaccine immunity, as challenge infection with in vivo-derived homologous inocula at 12 months after the first immunization provided no protection against in vivo-derived cell-free FIV challenge (17). In contrast, inactivated whole-virus vaccine has been reported to have longer duration of vaccine immunity than inactivated infected-cell vaccine (13, 18).
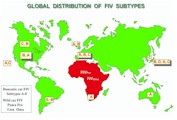 |
| Figure 1. [Enlarge] Global distribution of FIV subtypes. The subtypes are listed according to the order of predominance. Global sites included are Canada, United States, Hawaii, Argentina, United Kingdom, Europe, South Africa, Australia, Taiwan, and Japan. The occurrence of wild cat FIV in Africa is also presented. Results are compiled from references 3, 4, and 6. |
Inactivated whole-virus vaccines have provided the most consistent efficacy against homologous FIV challenges. A dual-subtype formulation of this vaccine was also effective against in vivo-derived heterologous FIV challenge (12, 13, 15). Conventional FIV vaccines have a unique advantage in that the viral antigens generally consist of closely related variants that develop in cell cultures, whereas molecular vaccine designs provide highly cloned viral antigens that are generally derived from a single variant. Due to the intrinsic diversity of variants in conventional vaccines, the immunity generated by these vaccines may be more expansive than that induced by molecular vaccines.
In order to further broaden vaccine immunity, two or three strains from different FIV subtypes were inactivated and analyzed individually and in combination with other strains against homologous and heterologous FIV challenges. FIV strains from FIV subtypes A (FIVPET) and D (FIVSHI) were determined to be the best combination, providing significant efficacy against homologous and heterologous in vivo-derived FIV challenges (15). Moreover, this vaccine protected cats against in vivo-derived challenge with subtype B envelope strain FIVBANG (15).
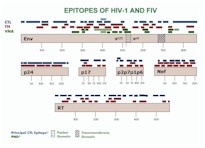 |
| Figure 2. [Enlarge] CTL, TH, and VNA epitopes of HIV-1 and FIV. CTL epitopes are shown in light blue and dark blue for FIV and HIV-1 respectively. TH epitopes are shown in maroon for HIV-1. VNA epitopes are shown in light green and dark green for FIV and HIV-1 respectively. Results are compiled from references 32-36. |
Another feature that makes this vaccine unique is that it is composed of FIV strains isolated from two long-term nonprogressor (LTNP) cats. An FIV-infected cat is considered to be LTNP when the animal remains clinically and hematologically asymptomatic for an extended period of time (at least 8 years). The rationale behind this selection process for vaccine strains is that less pathogenic strains (currently designated as LTNP) are more likely to generate stronger protective immunity than strains from rapid progressors and thus can serve as excellent vaccine immunogens. Additionally, the use of two subtypes focuses the immune response toward conserved epitopes that should, consequently, further broaden vaccine immunity across subtypes. Since the conceptualization of the dual-subtype LTNP vaccine, immunologic analyses of human LTNPs infected with HIV-1 suggest that they possess both broad-spectrum VNAs and cell-mediated immunity (CMI), including T-helper (TH) and cytotoxic T lymphocyte (CTL) responses, against HIV-1 (19-21).
An advantageous feature of the FIV AIDS model is that the FIV variants isolated from rapid progressor and LTNP cats can be tested for pathogenicity in the natural hosts, domestic cats. Based on experimental infection studies, the acute pathogenicity of five of the eight reported vaccine strains is presented in Table 1 along with the virus load and reported vaccine efficacy toward homologous challenge. The acute pathogenicity of the remaining three vaccine strains has not yet been published, but their relative virus load and vaccine efficacy have been reported and are presented.
Analysis of the pathogenicity of the vaccine strains clearly demonstrates that the prototype dual-subtype FIV vaccine strains have the nonpathogenic features of LTNP viruses. Recent studies have also demonstrated that the dual-subtype LTNP FIV vaccine induces broader VNAs and stronger CMI, including TH cytokine production (interferon g, interleukin-2) and cytotoxic responses (15) than single-subtype FIV vaccine administered at the same antigenic dose. Further, experimental FIV vaccines consisting of virulent or pathogenic FIV strains such as FIVGL8 and FIVBANG have generally been poor vaccine immunogens (22-24).
In light of the fact that the criteria for selection of vaccine strains have not been elucidated (25), the concept of using LTNP strains for vaccine immunogens is intriguing, although more studies with vaccines composed of pathogenic and nonpathogenic FIV strains are needed for verification. Worldwide testing of the dual-subtype LTNP FIV vaccine using the more natural setting of contact exposure to FIV-infected field cats will also address the ability of such vaccines to protect against natural isolates.
Concurrent advancement in challenge systems. As more experimental FIV vaccine trials were conducted, challenge systems that simulated natural infection became the standard for testing FIV vaccine designs. Inactivated whole-virus vaccine protection has been reported against in vivo-derived homologous challenge but not against in vivo-derived heterologous challenge (11). With the exception of one study, single-strain vaccines have not been greatly successful against challenge with moderately to greatly heterologous FIV strains (11, 14-16). To date, the majority of conventional inactivated single-strain FIV vaccines and molecular-based vaccines (plasmid DNA, recombinant vectored, recombinant peptide vaccines) have been either unsuccessful at protecting cats against challenge with heterologous in vivo-derived challenges or were not tested as such. Recent studies suggest that single-strain FIV vaccines may not be able to protect even against in vivo-derived homologous challenges (15, 26). In vivo-derived inocula contain quasi-species of FIV and simulate natural conditions more so than laboratory isolates that may have been partially "cloned" as the result of continuous passage in cell cultures.
This is in contrast to current vaccine designs in nonhuman primate models of AIDS vaccines where molecular technology has promoted the use of molecularly engineered challenge systems such as the pathogenic SHIV 89.6P. Current vaccine trial results using pathogenic SHIV as the challenge system have demonstrated disease prevention and amelioration of disease symptoms rather than prevention of infection, the latter commonly referred to as sterilizing immunity (27-29). Prior to the use of SHIV, only molecularly attenuated SIV vaccine provided both sterilizing immunity and disease prevention against wild-type SIV challenges. The attenuated vaccine approach is widely accepted when dealing with most viral infections of humans and animals. However, the nature of HIV-1 and FIV infection of their targeted hosts makes the use of an attenuated vaccine design less desirable or practical. The ability to infect and affect the immune cells, the same cells responsible for removing and controlling infections, is an important challenge to lentiviral vaccine design.
The foremost challenge raised by recent transmission studies on HIV-1 is the high rate of recombination between strains from the same and different subtypes. As many as 35% of the recent variants isolated from HIV-1-infected individuals are found to be recombinants of inter- and intrasubtype strains (25). Although the rate of recombination has not been determined for FIV, the existence of the inter- and intrasubtype recombinant FIV isolates (3, 24) and the relative ease in establishing dual infection in cats during experimental superinfection studies (30, 31) clearly support the view that FIV recombinants are prevalent in nature. Thus, the possibility that attenuated vaccine viruses may recombine with wild-type variants is the most important problem of the attenuated vaccine approach next to the possibility that molecular manipulations such as deletion of regulatory and auxiliary genes may not completely attenuate viruses without a major loss in the ability to confer protective immunity. The ability of current variants to superinfect the host raises yet another concern that not all HIV-1 and FIV strains may be useful as vaccine virus. Further, the pandemic nature of the HIV-1 and FIV infections as depicted by the distribution of various subtypes throughout the world (Figure 1) also presents a major challenge in vaccine design, suggesting that broadening vaccine immunity to include different subtype variants will require innovation as to approach and selection of the vaccine virus(es).
Insights to AIDS vaccine development
USDA approval of the dual-subtype FIV vaccine required that it induce sterilizing immunity against heterologous strains and that vaccine-induced immunity last at least 1 year. This first criterion has been most demanding, because the agency has previously approved veterinary vaccines that had significant efficacy at disease prevention in the absence of sterilizing immunity. A vaccine that only prevents disease without preventing infection raises a serious concern as to whether such a vaccine might "steer" the evolution of the AIDS lentiviruses to becoming more harmful to the host. Because AIDS drug therapy has advanced to the level of controlling the virus while providing the host an improved quality of life, the mission of vaccine prophylaxis should be to promote vaccines that induce sterilizing immunity. Thus, the rigorous policy set forth by the USDA appears to be the proper position to be taken by a federal regulatory agency.
Ideally, the antigenic composition of a vaccine should be based on the immunogenic epitopes on the viral proteins that provide protection against global isolates. Due to the limited data on specific FIV epitopes, the epitope analyses performed on HIV-1 have been used to lend insights into the development of both HIV-1 and FIV vaccines (Figure 2). Analysis of CTL, TH, and VNA epitopes of HIV-1 demonstrates that immunological epitopes are present throughout all of the viral structural proteins as well as on polymerases and regulatory proteins (Figure 2). If these epitopes can be considered to have equal immunological importance, then the presence of whole viral proteins is needed to induce appropriate immunity against the virus. The use of a conventional whole-virus vaccine is in keeping with this fundamental concept in that whole viral proteins are presented in natural form and as such may be important in inducing the appropriate prophylactic immunity against the virus. Whole-virus vaccines are capable of eliciting strong VNA and TH responses but are generally limited in CTL responses (15, 38-40).
Nevertheless, with better inactivation methods and more effective adjuvants including those that promote CTL responses, this vaccine approach may yet prove to be useful against AIDS viruses. The recent USDA approval of a dual-subtype FIV vaccine (Fel-O-Vax FIV, Fort Dodge Animal Health, Fort Dodge, Iowa) as a veterinary vaccine specifically for the prevention of FIV infection supports this concept. In the USDA-approved study, 67% (18/27 protection rate) of vaccinated cats were protected against heterologous virus challenge (at least 11% difference at Env), whereas 74% (25/34 infection rate) of control cats became persistently viremic. The viral status was based on both virus isolation and proviral PCR analysis of the DNA from leukocytes (13, 41). Thus, 1 year after initial vaccination, at least 67% of vaccinated cats had the proper protective immunity against FIV infection without additional boosting.
Analysis of the current vaccine designs in phase I through III clinical trials clearly demonstrates the concerted effort toward molecular-based vaccine designs (42). A similar trend has been noted in experimental FIV vaccine trials (Table 2). The resistance to using conventional vaccine approaches is based on the concern about the potential risk of incomplete inactivation of the vaccine virus. Although this risk factor is a serious concern and cannot be dismissed lightly, there is also an equal concern that the current molecular vaccine designs may not present sufficient levels of viral antigens in their most natural forms, thereby possibly not inducing the appropriate immunity against HIV-1 infection. This has been the case for current molecular FIV vaccines and has forced the FIV vaccine research community to also pursue the development of conventional vaccine approaches.
These efforts have resulted in the USDA-approved dual-subtype FIV vaccine. The development of this vaccine entailed the cooperation of academic institutions, industry, international organizations, and federal agencies. Even with such a partnership in place, vaccine development took 14 years of research at academic institutions and 10 years of effort by industry. Nevertheless, the pooling of these resources to develop a vaccine that can prevent the global spread of AIDS lentiviruses should be the central mission of all scientists and institutions involved.
References
| 1. | N. C. Pedersen, E. W. Ho, M. L. Brown, J. K. Yamamoto, Science 235, 790 (1987). PubMed |
| 2. | P. M. Sharp, et al., Phil. Trans. R. Soc. Lond. 356, 867 (2001). PubMed |
| 3. | M. H. Bachmann, et al., J. Virol. 71, 4241 (1997). PubMed |
| 4. | Y. Inoshima, et al., Arch. Virol. 143, 157 (1998). PubMed |
| 5. | S. Osmanov, et al., J. Acquir. Immune Defic. Syndr. 29, 184 (2002). PubMed |
| 6. | D. L. Sodora, et al., J. Virol. 68, 2230 (1994). PubMed |
| 7. | J. K. Yamamoto, et al., J. Am. Vet. Med. Assoc. 194, 213 (1989). PubMed |
| 8. | K. Hartmann, Vet. J. 155, 123 (1998). PubMed |
| 9. | R. E. Hill, "Issuance of Licenses," Center for Veterinary Biologics Notice No. 02-03 (USDA, Ames, IA, 2001). Available online |
| 10. | J. C. Watson, G. Peter, in Vaccines, S. A. Plotkin, W. A. Orenstein, Eds. (W.B. Saunders Company, Philadelphia, 1999), p. 51. [third edition] |
| 11. | J. Elyar, M.C. Tellier, J.M. Soos, J.K. Yamamoto, Vaccine 15, 1437 (1997). PubMed |
| 12. | E. W. Uhl, T. G. Heaton-Jones, R. Pu, J. K. Yamamoto, unpublished data. |
| 13. | "USDA issues license for feline immunodeficiency virus vaccine for cats," United States Department of Agriculture Animal and Plant Inspection Service press release (2002). Available online |
| 14. | M. J. Hosie, et al., J. Virol. 74, 9403 (2000). PubMed |
| 15. | R. Pu, et al., AIDS 15, 1225 (2001). PubMed |
| 16. | D. Matteucci, et al., J. Virol. 74, 10911 (2000). PubMed |
| 17. | D. Matteucci, et al., J. Virol. 71, 8368 (1997). PubMed |
| 18. | R. Pu, J. K. Yamamoto, unpublished data. |
| 19. | C. Rinaldo, et al., J. Virol. 69, 5838 (1995). PubMed |
| 20. | D. C. Montefiori, et al., J. Infect. Dis. 173, 60 (1996). PubMed |
| 21. | Y. J. Zhang, et al., J. Infect. Dis. 176, 1180 (1997). PubMed |
| 22. | M. J. Hosie, R. Osborne, G. Reid, J. C. Neil, O. Jarrett, Vet. Immunol. Immunopathol. 35, 191 (1992). PubMed |
| 23. | S. Finerty, et al., Vaccine 20, 49 (2002). PubMed |
| 24. | R. Pu, B. A. Torres, K. Phillips, M. Arai, J. K. Yamamoto, unpublished data. |
| 25. | P. Mooij, J. L. Heeney, Vaccine 20, 304 (2002). PubMed |
| 26. | W. Hesselink, P. Sondermeijer, H. Pouwels, E. Verblakt, C. Dhore, Vet. Microbiol. 69, 109 (1999). PubMed |
| 27. | M. I. Johnston, J. Flores, Curr. Opin. Pharmacol. 1, 504 (2001). PubMed |
| 28. | J. W. Shiver, et al., Nature 415, 331 (2002). PubMed |
| 29. | R. R. Amara, Science 292, 69 (2002). PubMed |
| 30. | S. Okada, R. Pu, E. Young, W. Stoffs, J. K. Yamamoto, AIDS Res. Hum. Retroviruses 10, 1739 (1994). PubMed |
| 31. | M. T. Kyaw-Tanner, W. F. Robinson, Arch. Virol. 141, 1703 (1996). PubMed |
| 32. | M. A. Rigby, et al., Vaccine 14, 1095 (1996). PubMed |
| 33. | J. Richardson, et al., J. Virol. 72, 2406 (1998). PubMed |
| 34. | J. N. Flynn, Immunology 85, 171 (1995). PubMed |
| 35. | J. N. Flynn, et al., J. Virol. 68, 5835 (1994). PubMed |
| 36. | S. D. Putney, et al., in AIDS Vaccine Research and Clinical Trials, D. P. Bolognesi, Eds. (Marcel Dekker, Inc., New York, 1990), pp. 25-33. |
| 37. | HIV Molecular Immunology Database, Epitope Maps, Los Alamos National Laboratory (2002). Available online |
| 38. | M. C. Tellier, J. M. Soos, R. Pu, D. Pollock, J. K. Yamamoto, Vet. Microbiol. 57, 1 (1997). PubMed |
| 39. | J. K. Yamamoto, et al., AIDS Res. Hum. Retroviruses 7, 911 (1991). PubMed |
| 40. | B. J. Willett, J. N. Flynn, M. J. Hosie, Immunol. Today 18, 182 (1997). PubMed |
| 41. | "Fort Dodge Animal Health received USDA approval for a feline immunodeficiency virus vaccine for cats," Fort Dodge Animal Health press release (2002). Available online |
| 42. | A. M. Schultz, J. A. Bradac, AIDS 15, S147 (2001). |
| 43. | This work was supported by grants NIH R01 AI30904 (JKY) and Miscellaneous Donor Fund (JKY). |
| 44. | Janet K. Yamamoto is the inventor of record on a University of Florida-held patent and may be entitled to royalties from companies that are developing commercial products that are related to the research described in this paper. |
| Copyright © 2001 by The American Association for the Advancement of Science |